Differential Scanning Calorimetry (DSC)
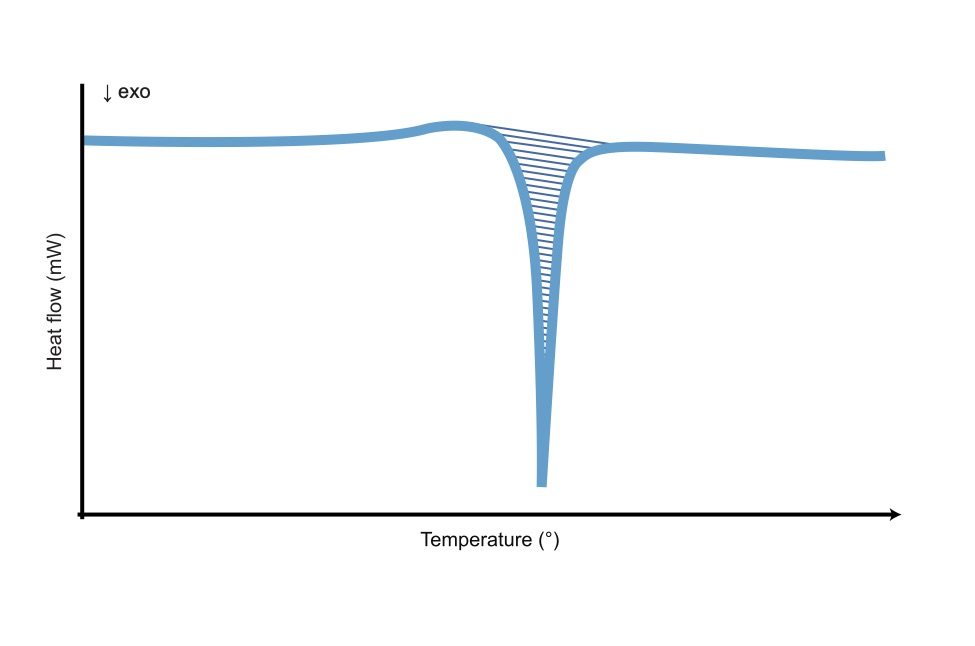
DSC is suitable to examine all physical processes taking place during heating and cooling of a pharmaceutical compound, for example, melting point, stability of the compound, decomposition temperature etc. Determination of the thermal behaviour is a required part of the physical characterisation of a pharmaceutical compound.
Differential scanning calorimetry is highly relevant to:
- Determine melting point, desolvation, glass transition, heat capacity, sublimation or thermal decomposition point of your compound, for instance, as an ID method.
- Examine the stability of the compound, as transitions or melting at low temperature is not desirable.
- Evaluate the relative stability of different crystalline forms to study transitions between states.
- Find out if the polymorphic forms are monotropic or enantiotropic.
- Evaluate crystallisation behaviour during heating – which might, for instance, lead to new polymorphic forms.
- Comparison of batches. Differences in energies upon melting might be due to amorphous material impurities or content, i.e. for evaluation of batch-to-batch variation.
- Purity evaluation by freezing point depression and applied for intermediates, raw materials, and final drug substances.
- Quantify and qualify the presence of the small amount of amorphous phase in a crystalline sample.
- Study stability of amorphous drug substances.
- Predict stability/instability of amorphous solid dispersions.
- Screen for co-crystals and polymorphs.
- Study phase diagram. Describe the type of enantiomer/stereoisomer/diastereomer mixture – compound or racemate.
- Study drug-excipient compatibility.
- Study oxidative stability by determining the oxidative-induction time (OIT) or oxidation-onset-temperature (OOT) of a sample and other chemical reactions.
- Perform safety screening—the presence of an exothermic event to assess the stability of a substance to heat.
- Study of retardation of the glass transition temperature as a function of relative humidity to predict the stability of amorphous materials and amorphous solid dispersion.
- Search for the best polymer for amorphous solid dispersion.
- The thermodynamics analysis of proteins can reveal necessary information about the global structure of proteins and protein/ligand interaction.
Instrument and measuring principle DSC
Measure the temperatures and heat flow associated with transitions in materials as a function of time and temperature in a controlled atmosphere.
A small amount of sample is needed. Differential scanning calorimetry measures the amount of energy (heat) absorbed or released when a specimen is heated, cooled, or held at a constant temperature.
The instrument applied by Particle Analytical is a Mettler DSC 823e Differential Scanning Calorimeter (DSC).
| Instrument | TA DSC2500 modulated DSC |
| USP/Ph. Eur. | USP 891/Ph. Eur 2.2.34 Thermal Analysis |
| Temperatures | -65 to 700 °C |
| Heating rate | Up to 20 °C/min |
| Sample amount | 5-10 mg |
Literature
Chiu MH, Prenner EJ (2011) Differential scanning calorimetry: An invaluable tool for a detailed thermodynamic characterisation of macromolecules and their interactions. J Pharm Bioallied Sci. 3(1):39-59.
Desai S, Shaikh M, Dharwadkar S (2003) Preformulation compatibility studies of etamsylate and fluconazole drugs with lactose by DSC. J Therm Anal. 71(2): 651-658.
O’Neill M J (1964) The Analysis of a Temperature-Controlled Scanning Calorimeter. Anal Chem 36(7): 1238–1245.
Pungor E, Horvai G, (2020) In A Practical Guide to Instrumental Analysis, CRC Press (15): 184-187.
Schön A, Brown RK, Hutchins BM, Freire E (2013) Ligand binding analysis and screening by chemical denaturation shift. Anal Biochem 443(1):52-57.
Verdonck E, Schaap K, Thomas LC (1999) A discussion of the principles and applications of Modulated Temperature DSC (MTDSC). Int J Pharm 192(1):3-20.



