X-Ray Diffraction (XRD)
X-ray diffraction (XRD analysis or XRPD analysis) is uniquely used for determination of crystallinity of a compound:
- I.D. of crystalline material (used for regulatory purposes or during development.)
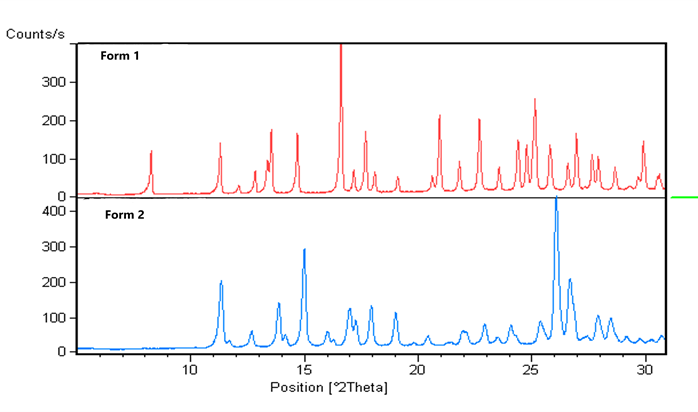
- I.D. of different polymorphic forms (“fingerprints” ).
- Distinguishing between amorphous and crystalline material
- Quantification of the percent crystallinity of a sample
- Quantification and qualification of polymorphic forms present in the sample
- Quantification and qualification analysis of crystalline chemical compounds present in the samples
- Analysis of crystalline structure of material
- Verification of co-crystal and crystalline salt formation
- Identify a solid-form of drug substance in a dosage form.
- Study buffer/solvent induced solid-form transition during dissolution and in suspensions
- Study stability of polymorphs in slurries.
- Conduct polymorph, salt, co-crystal screenings.
- Study phase diagrams for co-crystal scaling up of crystallisation process.
Knowledge about crystallinity is highly relevant, as a crystalline form is usually preferred in development: In contrast to amorphous material, a crystal has well-defined properties (melting point, solubility and IDR) – parameters that should be known in order to control your final product. The result from an XRD analysis is a diffractogram showing the intensity as a function of the diffraction angles. Positive ID of a material using XRD analysis is based on accordance between the diffraction angles of a reference material and the sample in question.
For Material Experts:
Instrument and measuring principle
XRD analysis is based on constructive interference of monochromatic X-rays and a crystalline sample: The X-rays are generated by a cathode ray tube, filtered to produce monochromatic radiation, collimated to concentrate, and directed toward the sample. The interaction of the incident rays with the sample produces constructive interference (and a diffracted ray) when conditions satisfy Bragg’s Law (nλ=2d sin θ). This law relates the wavelength of electromagnetic radiation to the diffraction angle and the lattice spacing in a crystalline sample.
The characteristic x-ray diffraction pattern generated in a typical XRD analysis provides a unique “fingerprint” of the crystals present in the sample. When properly interpreted, by comparison with standard reference patterns and measurements, this fingerprint allows identification of the crystalline form.
| Instrument | PANalytical X’Pert³ Powder and Multipurpose X-ray Diffractometer Empyrean |
| USP/Ph. Eur. | Ph. Eur 2.9.33 |
| Operating range | 2-60 °2Theta (if required up to 150 °2Theta) |
| Temperature | R.T. (temperature chamber available) |
| Humidity | Ambient (humidity chamber available) |
| Sample amount | App. 10 mg-200 mg |



